About Us

【TRANSWELL BIOTECH: Advancigenerative
Medicine, Leading the Future of Bio-Innovation】
Established in 2010 and headquartered in the Hsinchu Science Park, Taiwan, TRANSWELL BIOTECH is an innovative biopharmaceutical company dedicated to regenerative medicine. TRANSWELL possesses cutting-edge cell and tissue engineering technologies and facilities, featuring international-standard Class 10,000 cleanrooms and comprehensive QC analytical laboratories. These ensure that every stage of our cell products, from R&D and manufacturing to storage, is maintained within a strictly rigorous and controlled environment.
We are committed to pioneering R&D in high-end biological wound dressings, cell therapies, exosome technology, medical devices, and serum-free culture media, continuously transforming advanced biomedical science into medical solutions with significant clinical value.

【Core Competencies: International Partnerships and
Proprietary Technologies】
TRANSWELL BIOTECH collaborates with leading international biomedical institutions and has secured global exclusive licenses from the University of Lausanne, Switzerland, and ELANIX, granting access to a GMP-compliant allogeneic dermal fibroblast cell bank. In Taiwan, we scale up and optimize cell manufacturing processes at ITRI’s GTP laboratory, implementing standardized procedures and stringent quality control to ensure high product purity and stable biological activity.
We successfully developed Taiwan’s first allogeneic cell therapy product, overcoming critical technical barriers. This achievement highlights TRANSWELL’s core competencies in cell manufacturing and regenerative medicine, laying a solid foundation for product commercialization and clinical application.
【TWB-103 Allogeneic Dermal Fibroblast Cell Therapy】
TWB-103 is an allogeneic cell therapy product developed by TRANSWELL BIOTECH, specifically designed for the repair of acute and chronic wounds. The product combines dermal fibroblasts stored at -80°C with a 4°C hydrogel carrier, offering long-term preservation and a “ready-to-use” feature for rapid clinical application. Upon application, the cells are mixed and applied directly to the wound, where they continuously release multiple growth factors to promote cell proliferation, angiogenesis, and tissue regeneration, while improving the wound microenvironment. The hydrogel carrier maintains cell viability, provides an optimal moist environment, and reduces pain and discomfort during dressing changes, enhancing the overall treatment experience.
By integrating innovative technology with clinical practicality, TWB-103 offers a faster, more effective, and novel solution for advanced wound care.

【EpiV Serum-free Epithelial Cell Culture Medium】
EpiV Medium is a chemically-defined, professional-grade culture medium that is serum-free, BPE-free, HPL-free, and cholera toxin-free. Designed for feeder-free systems, it is specifically formulated for the culture of epithelial cells and keratinocytes. The medium effectively suppresses fibroblast growth, provides an optimal adhesion environment, and promotes cell proliferation.
Traditional keratinocyte culture is challenging, as most conventional media contain bovine pituitary extract (BPE), which poses unknown composition and BSE risks, limiting clinical applications. BPE-free alternatives often suffer from inadequate culture performance. EpiV Medium, licensed from the Medical and Pharmaceutical Industry Technology and Development Center (PITDC) and optimized by TRANSWELL BIOTECH, demonstrates superior cell adhesion, proliferation efficiency, and passage number compared to competing products, making it an ideal choice for large-scale clinical cell culture.
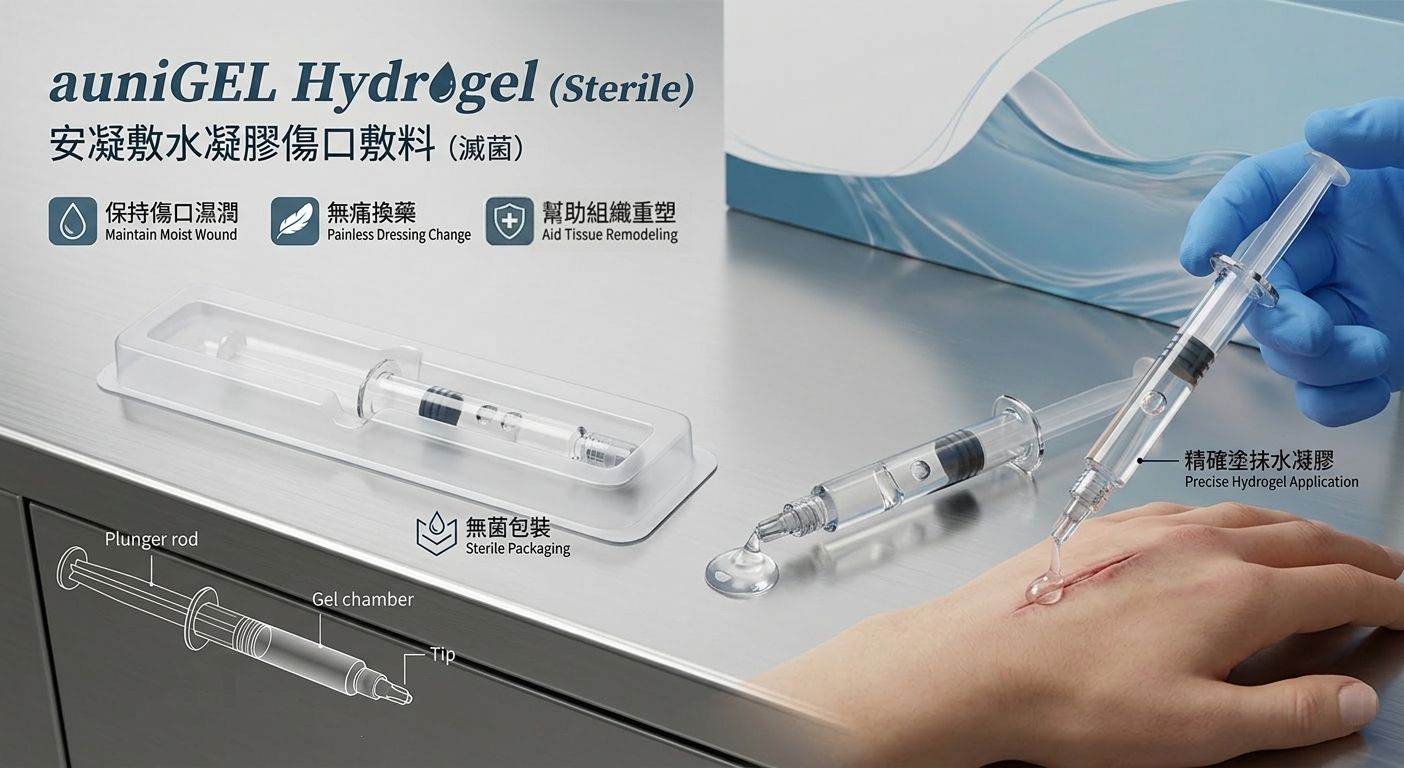
【auniGEL Hydrogel Wound Dressing】
auniGEL is a sterile hydrogel wound dressing designed for trauma, post-laser treatment, burns, and superficial wounds, suitable for low-exudate, non-infected areas. After cleaning and disinfecting the wound, the dressing is evenly applied to maintain a moist environment, promoting natural healing while enhancing user comfort.
The product comes in single-use sterile packaging and can be stored at room temperature. Prior to use, it can also be cooled at 4°C to increase viscosity and improve application. Classified as a Class II medical device (Taiwan FDA Registration No. 008424), auniGEL combines safety and convenience, making it an ideal choice for everyday wound care.

TWB-103
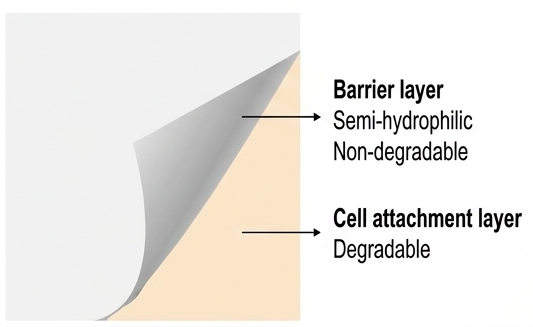
TWB-301

EpiV Medium

auniGEL Hydrogel

Exosomes/EVs

